- Expertise
- Services
- Solutions
- About Us
About Us
Overview
Leadership
Facility and Equipment
Quality and Compliance
Therapeutic Areas
Why Us?
Values
Process
Partners
Careers
- Business Development Associate
- Sr. Scientist – Cell Based Assay
- GLP Quality Assurance Manager
- Sr. Scientist, ELISA Immunoassay Bioanalysis
- Sr. Scientist, LC-MS/MS Bioanalysis
- Associate Scientist, Central Laboratory Services
- Sr. Scientist - Pharmacokinetics (PK)
- Content Manager, Scientific and Regulatory Writing
- Resources
- Request Quote
TaqMan Gene Expression Assay, Copy Number Variation Assay Development, nGLP Validation, And Sample Analysis

Challenge
To develop a reliable and sensitive gene expression analysis for the pharmacodynamic biomarkers to respond to the therapeutic drug treatment. Real-time PCR gene expression, or qRT-PCR gene expression, or qPCR analysis of gene expression was conducted on multiple members of the Wnt/β-catenin signaling pathway and their downstream targets. Specifically, differential gene expression analysis, qPCR expression analysis, and ddPCR gene expression were conducted on differentially expressed genes such as Wnt Family member 16 (Wnt16), Lymphoid Enhancer-Binding Factor (LEF1), Melanogenesis Associated Transcription Factor (MITF), SRY-box 10 (SOX10), adhesion markers β-catenin-1 (CTNNB1), Cadherin 1 (CDH1), and the proteases Matrix Metalloproteinase-9 (MMP-9). Gene expression profiling also included multiple housekeeping genes such as RNA polymerase II subunit A (POLR2A, Eukaryotic 18S rRNA (18S), Actin-beta (ACTB), and Ribosomal Protein Lateral Stalk subunit P0 (RPLP0) were analyzed as a control gene. Some of these differentially expressed genes exhibit copy number variation and low expression levels, presenting challenges in precise quantification. Therefore, differential gene expression analysis, copy number analysis, and copy number variation assay were conducted to ensure accurate quantification of gene expression levels and account for any copy number variations across samples.
Solution
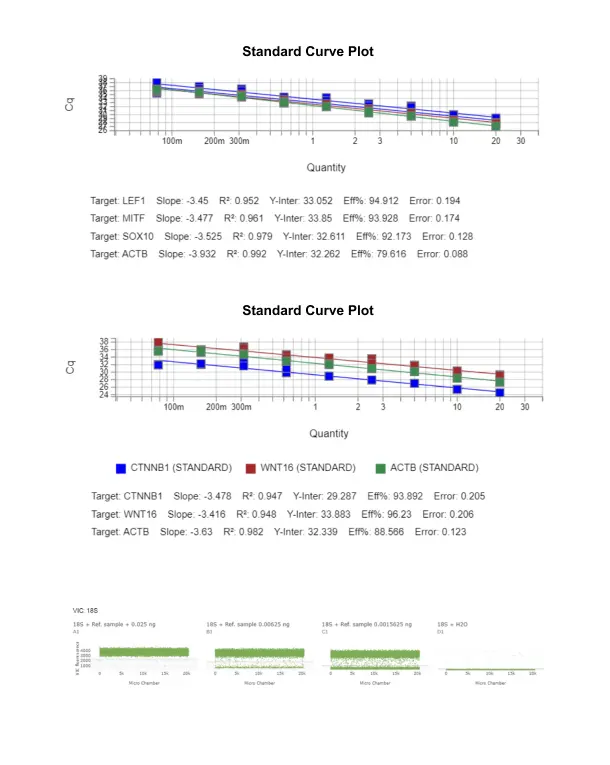
A TaqMan Gene Expression Assay was used to optimize and quantify the expression levels of these differentially expressed genes in limited amounts of human biological matrices obtained by biopsy. Because of the limited sample quantity and wide variation in the expression of the target and housekeeping genes, careful and thorough optimization was first carried out on quantitative real-time PCR gene expression (qPCR) before quantification of gene expression on the digital PCR (ddPCR) gene expression platform.
This intricate optimization method involves setting up qRT-PCR gene expression experiments with serially diluted complementary DNA (cDNA) to generate a standard curve to evaluate primers/probes’ amplification efficiencies, a crucial step in qPCR expression analysis. Overall, the precision in qPCR analysis of gene expression is vital for ensuring the reliability of our findings in this limited-sample study. This approach is crucial for accurate copy number analysis and copy number variation assay, especially when dealing with limited sample quantities and copy number variations in gene expression levels. This process is essential for analyzing differentially expressed genes and conducting gene expression profiling, ensuring precise and reliable results in differential gene expression analysis.
To overcome the limited RNA amount, we leveraged dPCR (QuantStudio Absolute Q Digital PCR System) for gene expression analysis of differentially expressed genes. Unlike qRT-PCR gene expression analysis, which relies on real-time PCR gene expression technology, ddPCR gene expression involves partitioning a sample into real-time PCR gene expression reactions. Some reactions contain the target molecule (positive), while others do not (negative). To ensure a seamless transition from qPCR to ddPCR, the initial qPCR analysis of gene expression established a robust foundation for precise measurements in our experiments. After PCR amplification is completed, the negative fraction is used to generate an absolute quantity for the exact number of target molecules in the sample on a digital PCR platform using Poisson statistics and analysis. This method facilitates differential gene expression analysis and copy number analysis, offering several advantages over qPCR expression analysis. Unlike qPCR, ddPCR gene expression does not require known standards or references, and results are presented as copy numbers, aiding in gene expression profiling and copy number variation assay.

Outcome
We developed a reliable and transferable TaqMan Gene Expression Assay method for gene expression analysis of targets in a limited biological matrix such as skin biopsy. Following optimization on qPCR expression analysis, real-time PCR gene expression assays were used to quantify the copy number of targets and housekeeping genes, enabling precise copy number analysis. This approach facilitates the identification of differentially expressed genes and enables gene expression profiling, allowing for comprehensive analysis of genetic variations and their implications.
Since gene expression levels (biomarkers) have proven to be of high diagnostic and therapeutic value, qRT-PCR gene expression analysis is essential. The ddPCR gene expression platform is emerging as a more valuable tool for analyte quantitation and robust bioanalysis of multiple biomarkers with high accuracy and precision using only a small sample quantity or volume. This makes it ideal for differential gene expression analysis, copy number analysis and copy number variation assay in diverse biological samples.
Speak To Our Scientist About Your Quantitative PCR and Digital PCR TaqMan Gene Expression Assay Development, nGLP Validation, And Sample Analysis
Similar Case Study Articles
-

Multiplex Cytokine Assay, GLP Cytokine Validation, Cytokine Quantification On MSD
-

Humanized Monoclonal Antibody (MAb) PK ELISA Development, GLP Validation, And MAb Bioanalysis
-

Oligonucleotide Hybridization ELISA PK Assay, GLP Validation, Sample Analysis
-

Genomic Variation Analysis – TaqMan Single Nucleotide Polymorphism (SNP) Analysis
-

LC MS Bioanalysis Of Oligos Using Mass Spec And Ion Pair Chromatography
-

Dosing THC Lab, Cannabis Testing Lab, CBD Melatonin Testing
-

HPLC-UV Method Development: Testing 9 Articles in Formulation
-

Payload Plus Peptide LC MS Analysis, GLP Validation In Rat, NHP, Human Plasma
-

Epigallocatechin gallate (EGCG) LC MS Analysis In VcEDTA Human Serum
-

LC MS Method For Proline Amino Acid, Hydroxyproline In Mouse Tissues
-

LC MS Method For DNA Adducts Analysis In Cell-Based In-Vitro Samples
-

LC MS Method for Everolimus Eluting Coronary Stent Drug Delivery Systems
-

Mass Spectrometry Amino Acids, Quantitative LC MS Amino Acid Analysis
-

LC MS Method Development And GLP Validation For PK Plasma Sample Analysis
-

Incurred Sample Reanalysis (ISR) Test, FDA Requirements, And Study Evaluation
-

Dried Blood Spot (DBS) Sampling, DBS Collection, And DBS Analysis