- Expertise
- Services
- Solutions
- About Us
- Resources
- Request Quote
Humanized Monoclonal Antibody (MAb) PK ELISA Development, GLP Validation, And MAb Bioanalysis
Challenge
A biotech sponsor approached us for the development and GLP validation of a PK ELISA specific to their MAb antibody bioanalysis needs. They aimed to quantify a humanized mAb fragment (FAb) targeting a growth factor implicated in a prevalent age-related degenerative disease. The process of MAb ELISA development entails several critical considerations to ensure robust bioanalytical outcomes.
Humanized MAb fragments offer distinct advantages over whole antibodies, including lower production costs, enhanced tissue penetration, and minimized risk of triggering unintended immune responses. However, these fragments pose unique challenges for immunoassay design due to the absence of domains like the Fc region, which are commonly utilized for binding and detection in immunoassays. This can affect the sensitivity of MAb ELISAs, as the binding capacity of each fragment is reduced. Despite these challenges, precise MAb PK profiling is vital for delineating the drug’s pharmacological profile.
To navigate these complexities, meticulous planning of critical reagents, alongside strategic MAb ELISA development and GLP validation, is essential. While optimizing reagent concentrations could potentially enhance assay sensitivity, it also risks amplifying background noise from both specific and nonspecific interactions among assay components, such as the therapeutic molecule, blocking agents, and coating antigen. This is particularly problematic when the assay matrix is derived from a species closely related to humans, like non-human primates, due to the possibility of cross-reactivity.
The development and validation of MAb immunoassays, therefore, demand careful attention to the unique characteristics of MAb fragments, fine-tuning of assay elements for heightened sensitivity and specificity, and the mitigation of cross-reactivity risks.
With the right MAb ELISA approach, the detection and quantification of monoclonal antibodies across diverse samples can be significantly improved. Conducting comprehensive MAb PK studies is indispensable for understanding the in vivo distribution and behavior of these antibodies. Through diligent MAb development and bioanalysis, we can achieve the assay performance necessary to fulfill the sponsor’s requirements for assessing the therapeutic impact of the MAb fragment. This holistic strategy is pivotal in propelling MAb therapeutics forward, ensuring their clinical efficacy and safety.
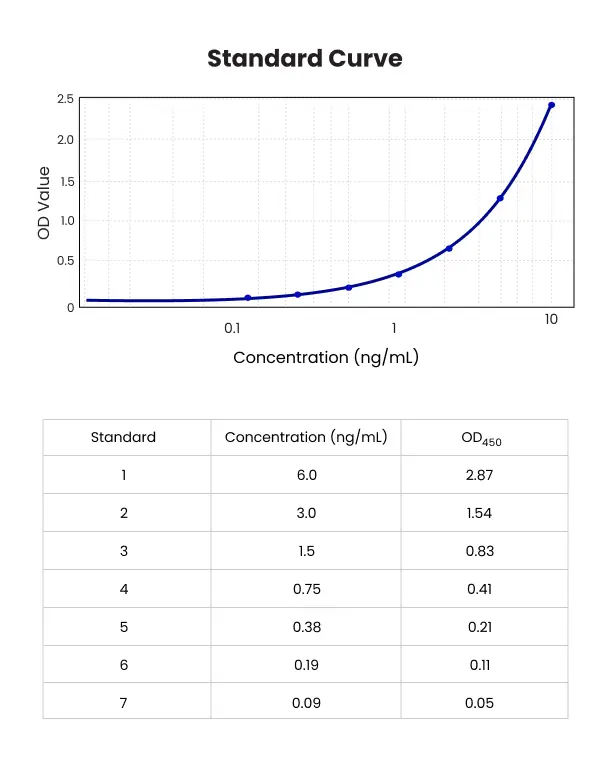
Solution
Our team leveraged decades of collective experience in immunoassay development and troubleshooting to explore various assay configurations for MAb development. We concentrated on refining the assay for a humanized MAb to ensure precise detection and analysis.
We determined that a robust and reproducible indirect ELISA was the best approach for assaying this therapeutic. This setup involved using the antigen target of the monoclonal antibody fragment for capture and a secondary antibody against the MAb’s Fab fragment for detection. Despite its effectiveness, we faced challenges with increased background noise due to detecting only a portion of the MAb antibody.
To address this, we employed a checkerboard approach to find the optimal concentrations for coating and detection antibodies. Our veteran team also refined several aspects of the initial immunoassay, introducing a novel method that involved preincubating the detection antibody with a small percentage of a homologous matrix. This innovation significantly improved the immunoassay’s signal-to-noise ratio and sensitivity, enhancing both GLP validation and sample analysis for MAb bioanalysis.
The optimized assay enabled accurate pharmacokinetic (PK) profiling, which is crucial for understanding MAb PK behavior. Our rigorous MAb bioanalysis protocols ensured precise assessment of the MAb therapeutics’ efficacy and safety profiles. Through meticulous optimization and validation, we secured reliable data essential for advancing the MAb development of this important humanized MAb therapeutic.

Outcome
An immunoassay meeting the sponsor-mandated sensitivity and specificity requirements was successfully developed and validated according to FDA bioanalytical guidelines. The bioanalysis of MAb antibody was completed in plasma and ocular fluid matrices for the GLP pharmacokinetic (PK) study in non-human primates (NHPs). Additionally, the MAb ELISA method was employed to ensure precise quantification and validation in these matrices.
Over the past decade, MAb antibody, well-known for its use in immunodetection-based assays, has also emerged as therapeutic agent for treating various diseases. Humanized MAbs, in particular, have gained prominence due to their reduced immunogenicity and improved efficacy in human patients. To provide differentiated therapeutic advantages or overcome side effects, researchers are increasingly developing modified MAb antibody fragments and seeking FDA authorization. Incorporating MAb PK studies is critical in these evaluations, as they provide detailed pharmacokinetic profiles essential for regulatory submissions. Thus, novel approaches to assay these MAb therapeutics, building on traditional immunoassay methods, are highly valuable for facilitating regulatory approval.
Advancements in MAb development and bioanalysis streamline the approval process and ensure the safety and efficacy of new treatments, significantly contributing to healthcare progress. Techniques such as MAb ELISA and MAb PK studies are crucial for accurately assessing therapeutic efficacy and safety profiles. By employing state-of-the-art methodologies, researchers gain deeper insights into the pharmacokinetics and pharmacodynamics of monoclonal antibody fragments, leading to more informed clinical decisions and regulatory submissions. Furthermore, robust MAb bioanalysis supports the exploration of novel MAb therapeutic modalities and optimization of treatment strategies, ultimately benefiting patients and advancing the field of medicine.
Speak To Our Scientist About Your Monoclonal Antibody Fragment PK ELISA Immunoassay Development, GLP Validation, and MAb Bioanalysis
Similar Case Study Articles
-

Multiplex Cytokine Assay, GLP Cytokine Validation, Cytokine Quantification On MSD
-
Oligonucleotide Hybridization ELISA PK Assay, GLP Validation, Sample Analysis
-

TaqMan Gene Expression Assay, Copy Number Variation
-

Genomic Variation Analysis – TaqMan Single Nucleotide Polymorphism (SNP) Analysis
-
LC MS Bioanalysis Of Oligos Using Mass Spec And Ion Pair Chromatography
-
Dosing THC Lab, Cannabis Testing Lab, CBD Melatonin Testing
-
HPLC-UV Method Development: Testing 9 Articles in Formulation
-
Payload Plus Peptide LC MS Analysis, GLP Validation In Rat, NHP, Human Plasma
-
Epigallocatechin gallate (EGCG) LC MS Analysis In VcEDTA Human Serum
-
LC MS Method For Proline Amino Acid, Hydroxyproline In Mouse Tissues
-
LC MS Method For DNA Adducts Analysis In Cell-Based In-Vitro Samples
-
LC MS Method for Everolimus Eluting Coronary Stent Drug Delivery Systems
-
Mass Spectrometry Amino Acids, Quantitative LC MS Amino Acid Analysis
-

LC MS Method Development And GLP Validation For PK Plasma Sample Analysis
-

Incurred Sample Reanalysis (ISR) Test, FDA Requirements, And Study Evaluation
-
Dried Blood Spot (DBS) Sampling, DBS Collection, And DBS Analysis