- Expertise
- Services
- Solutions
- About Us
About Us
Overview
Leadership
Facility and Equipment
Quality and Compliance
Therapeutic Areas
Why Us?
Values
Process
Partners
Careers
- Business Development Associate
- Sr. Scientist – Cell Based Assay
- GLP Quality Assurance Manager
- Sr. Scientist, ELISA Immunoassay Bioanalysis
- Sr. Scientist, LC-MS/MS Bioanalysis
- Associate Scientist, Central Laboratory Services
- Sr. Scientist - Pharmacokinetics (PK)
- Content Manager, Scientific and Regulatory Writing
- Resources
- Request Quote
Precision HPV Quantification: TaqMan qPCR/dPCR Method Development and Clinical Sample Analysis

Challenge
High-risk human papillomavirus (HPV) genotypes—most notably HPV16 and HPV18—account for the majority of HPV-attributable cervical cancers and are central drivers of cervical carcinogenesis through sustained expression of the viral oncogenes E6 and E7. In contrast, HPV11 is considered a low-risk genotype, most commonly associated with benign anogenital warts and recurrent respiratory papillomatosis rather than cervical carcinoma; it is sometimes included in assay panels as a clinically relevant comparator or specificity control.
As cervical disease progresses from transient infection to persistent infection and, in some cases, high-grade lesions and invasive cancer, the measurable HPV DNA burden can change substantially. In this context, detecting true biological shifts can be challenging with conventional quantitative real-time PCR (qPCR) when target abundance is low or near the assay’s limit of quantification.
Solution
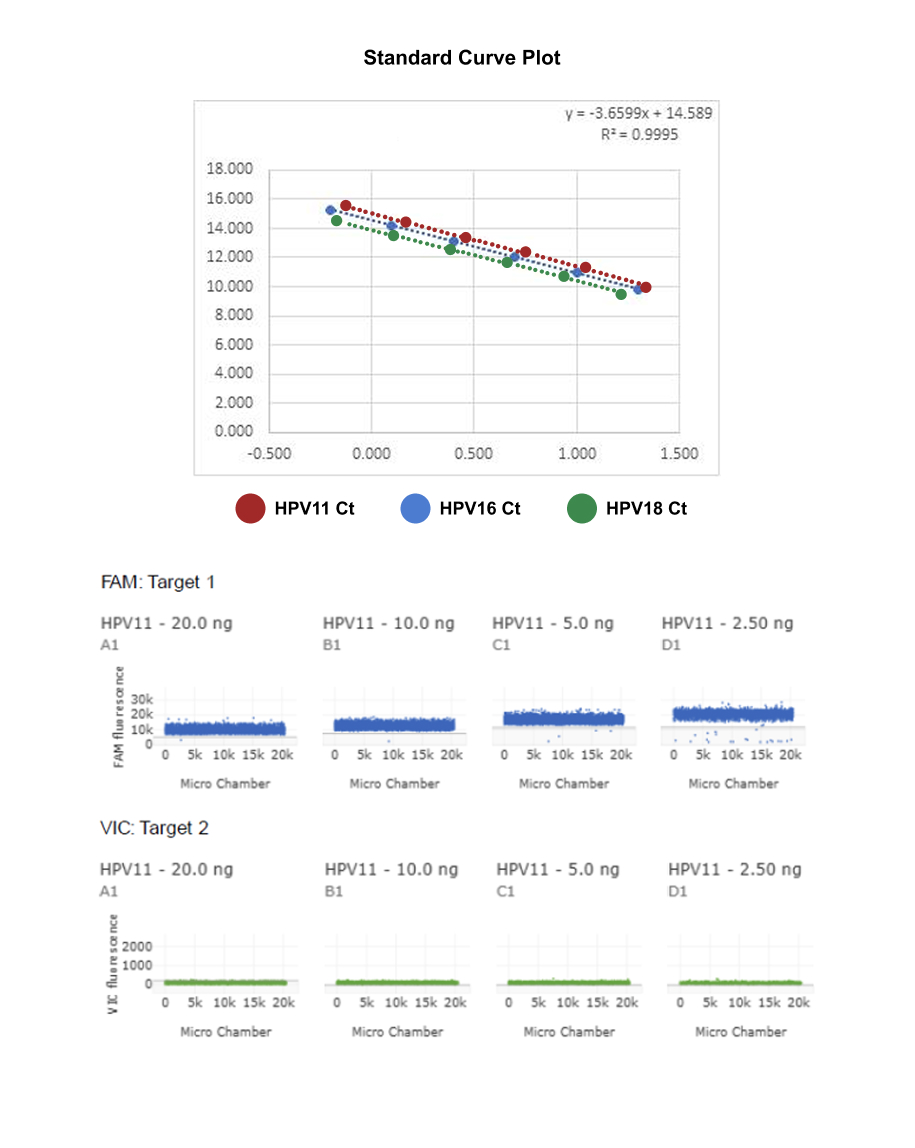
A TaqMan Gene Expression Assay was used to optimize and quantify HPV11, HPV16, and HPV18 targets in human biological matrices (whole blood or plasma), where sample volume is limited and target abundance can span a wide dynamic range. To maximize sensitivity and reproducibility, we first performed a rigorous optimization on the quantitative real-time PCR (qPCR) platform before transferring the assay to digital PCR (dPCR) for final quantification.
Method optimization began with qPCR experiments using serial dilutions of quantified HPV plasmid DNA to generate standard curves. These data were used to confirm primer/probe performance, assess amplification efficiency, and establish the limit of detection (LOD)—critical prerequisites for reliable qPCR-based quantification, particularly when copy number is low and copy number variation may influence apparent signal.
To address the constraints of limited DNA input and improve precision at low target levels, we then leveraged dPCR (QuantStudio Absolute Q Digital PCR System). In dPCR, each sample is partitioned into thousands of individual PCR microreactions, such that some partitions contain the target (positive) and others do not (negative). After endpoint amplification, the fraction of negative partitions is used to calculate the absolute target concentration (copies per input volume) using Poisson statistics, enabling direct reporting in copy number units.

Outcome
This workflow provides a smooth transition from qPCR to dPCR: qPCR establishes robust assay performance (efficiency, sensitivity, specificity), while dPCR delivers absolute quantification without a standard curve, improved tolerance to variability near the lower range, and clearer resolution for gene expression profiling, copy number analysis, and copy number variation (CNV) assessment in complex human matrices.
To address these limitations, we developed a sensitive and robust digital PCR (dPCR) assay capable of simultaneously detecting and accurately quantifying multiple HPV genotypes. Performance was benchmarked against real-time qPCR using serially diluted HPV11, HPV16, and HPV18 plasmid DNA standards, with water/no-template controls included throughout to monitor background and contamination.
Across the same input DNA samples, dPCR consistently delivered superior precision and sensitivity, with excellent specificity—particularly in the low-abundance range where qPCR measurements can become variable and less dependable.
Speak To Our Scientist About Your Multiplex Cytokine Assay, Cytokine Quantification On Meso Scale Discovery
Similar Case Study Articles
-
Humanized Monoclonal Antibody (MAb) PK ELISA Development, GLP Validation, And MAb Bioanalysis
-

Oligonucleotide Hybridization ELISA PK Assay, GLP Validation, Sample Analysis
-

TaqMan Gene Expression Assay, Copy Number Variation
-

Genomic Variation Analysis – TaqMan Single Nucleotide Polymorphism (SNP) Analysis
-

LC MS Bioanalysis Of Oligos Using Mass Spec And Ion Pair Chromatography
-

Dosing THC Lab, Cannabis Testing Lab, CBD Melatonin Testing
-

HPLC-UV Method Development: Testing 9 Articles in Formulation
-

Payload Plus Peptide LC MS Analysis, GLP Validation In Rat, NHP, Human Plasma
-

Epigallocatechin gallate (EGCG) LC MS Analysis In VcEDTA Human Serum
-

LC MS Method For Proline Amino Acid, Hydroxyproline In Mouse Tissues
-

LC MS Method For DNA Adducts Analysis In Cell-Based In-Vitro Samples
-

LC MS Method for Everolimus Eluting Coronary Stent Drug Delivery Systems
-

Mass Spectrometry Amino Acids, Quantitative LC MS Amino Acid Analysis
-

LC MS Method Development And GLP Validation For PK Plasma Sample Analysis
-

Incurred Sample Reanalysis (ISR) Test, FDA Requirements, And Study Evaluation
-

Dried Blood Spot (DBS) Sampling, DBS Collection, And DBS Analysis