What is LC-MS (Liquid Chromatography Mass Spectrometry)?
LC mass spectrometry is an analytical technique that combines chromatographic separation of target compounds (or analytes) followed by their mass-based detection. The sensitivity, selectivity, and accuracy especially of liquid chromatography tandem triple-quadrupole mass spectrometry (LC-MS/MS) has resulted in widely utilized procedures optimal for detecting nanomolar or even picomolar quantities of various analytes ranging from drugs, drug and food metabolites, biomarkers of disease progression or drug efficacy, pesticides, food contaminants, markers of ecosystem stability, and natural product extracts.
Understanding LC-MS
Nowadays, the most widely utilized bioanalytical method for quantitation is LC-MS/MS, or liquid chromatography (LC) tandem triple-quadrupole mass spectrometry (MS/MS). The LC/MS/MS procedure is similar to HPLC/UV with regards to sample preparation and chromatographic setup. Compared to UV detection, however, MS/MS offers superior sensitivity and selectivity, allowing for higher throughput analyses with more sensitive lower quantitation limits. LC/MS/MS is a beneficial, robust, and sensitive procedure used for a wide variety of small molecules. Furthermore, this technology is amenable to automation and unattended analysis.
Mass Spectrometry Schematic
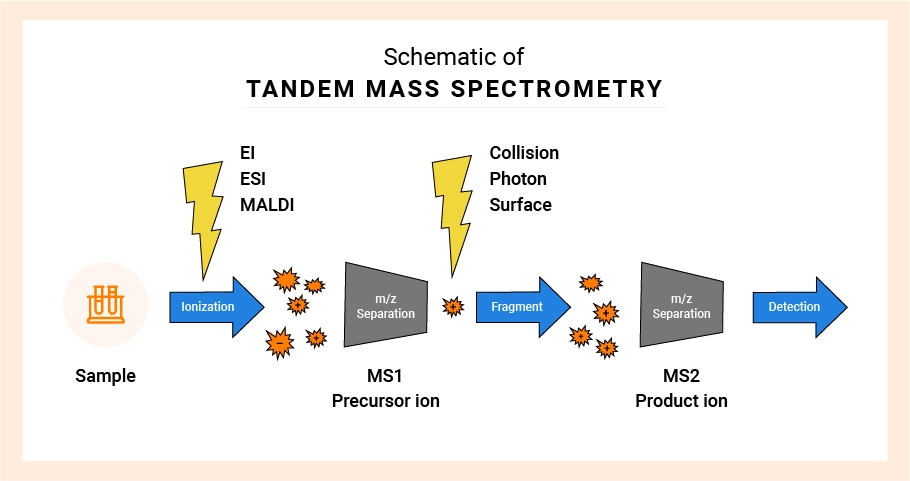
LC-MS/MS operates liquid chromatography in a parallel method to that described for HPLC. Triple quadrupole mass spectrometry is a detection technique that depends on the mass-to-charge ratio of a precursor ion and a corresponding product ion for each analyte of interest. Following the chromatographic separation, molecules are ionized via source energy, typically by electrospray ionization (ESI) or atmospheric-pressure chemical ionization (APCI). These ionized molecules then go through a set of charged rods, or a quadrupole Q1 in a triple-quadrupole instrument, separating particles based on their mass-to-charge ratio. These ions then pass through the second quadrupole (Q2), also called the collision cell. Following collision and fragmentation, product ions are then filtered through Q3 based on their mass-to-charge ratios for selective detection based on the parent and product ion pairings creating a unique MRM transition for each detected compound in a given assay. Detection is performed based on charged atoms reaching a detector such as an electron multiplier and creating current. The detector only detects charges and cannot differentiate based on mass. The magnetic separation provides the selectivity of the methodology, driven through software that regulates and monitors changes in rapid timeframes. Thus, two molecules that are ionized simultaneously but have different masses can be detected independently using MS detection. Similarly, two molecules that are ionized simultaneously and have different masses but have additional charges can be seen separately using MS detection.
Additional specificity can be found when multiple ionization procedures occur. The MS/MS technology is first used when a single ionization is insufficient to achieve the desired sensitivity. The subsequent ionization process fragments the molecules into fragments with smaller mass-to-charge ratios, with each fragmentation resulting in a charged (detectable) product ion and an uncharged (undetectable) neutral loss component. Each parent molecule fragments in a predictable manner based on the compound’s structure. Thus, even if two-parent molecules have the same mass-to-charge ratio, they can differentiate if they break into differentiated productions. The multiple ionization event methods are termed tandem mass spectrometry.
Liquid Chromatography separation
Tandem Mass Spectrometry
The specificity of mass spectrometry detection permits more flexibility and efficiency with liquid chromatographic separations. Because MS/MS can uniquely detect molecules with differentiable products, even if they co-elute, the liquid chromatography separation becomes less critical for LC/MS/MS than HPLC/UV as long as quantitative selectivity can be demonstrated. Often, simple chromatographic methods are adequate for LC/MS/MS, where complete separation is required for HPLC/UV. The result is that run times can be much shorter with LC/MS/MS, and precision and accuracy are increased. It is the best of both worlds!
The prime challenge in MS detection is controlling the ionization process. Problems with incomplete ionization and interference can lead to poor precision and accuracy with the MS detector. These problems can be resolved by changing detection, various ionization techniques, sample preparation solvents, LC stationary phases (columns), or LC mobile phases, and chromatographic separations by isocratic or gradient methodologies. Additional detection techniques include a time-of-flight MS, ion capture MS, and others. Source ionization techniques include electrospray ionization (ESI), atmospheric pressure chemical ionization (APCI), photoionization, or other specialized techniques. These unique methods use different MS detector setups to improve the precision, accuracy, selectivity, and sensitivity of MS detection. Additionally, due to the necessity for minimizing introduction of non-volatile components to the MS source, LC buffer salt choices are limited to volatile buffer systems (typically ammonium cations with formate or acetate anions) and may offer limited buffering capacity at typical mobile phase pH levels.
It is unlikely that you will be skilled in mass spectrometry after reading this article; but we hope to at very least to impart an overview of the versatility of the technique and its application to sample analysis and bioanalysis.
Drug development is carried out in humans and animals worldwide; however, as computing ability and the number of sophisticated technology platforms grows exponentially, our data on human health and disease increases.
- Partition chromatography
- Ion-exchange chromatography
- Size-exclusion chromatography
- Affinity chromatography
Basics of Liquid Chromatography
The mobile phase flows through the column and a detector that “responds” to the analytes’ firm physical or chemical properties, for instance, light absorption or emission, refractive index, or mass-to-charge ratios.. This response is captured as a peak or signal whose peak height or area (intensity) matches the total amount of the component available in the sample. The time the detector “sees” the analyte is its retention time (RT). An analyte’s uniqueness may be confirmed by comparing RT with the RT of a known standard, and ensuring blank samples show no measurable signal and that signals increase in a predictable fashion with respect to concentration. While this is not a precise method of compound identification, it helps when minimal data about a sample is known a priori.
Using Mass Spectrometry Detection with Liquid Chromatography Separation
Even though many detectors of differing sensitivity and technologies have been coupled with LC for analyzing different sample types, the mass spectrometer (MS) has emerged as a selective, sensitive, and widely universal detector offering improved selectivity, sensitivity, and efficiency.
While several detector types allow for flow-through analysis, the LC eluent carrying the analytes cannot directly flow into the mass spectrometer. The LC MS system and mass spectrometers are coupled through an interface (or ion source), and LC is operated at normal back-pressures. The mass spectrometer detector is operated under a vacuum. As the column eluent flows, the solvent is ionized or evaporated by applying voltage and heat. The charged analyte molecules are then introduced into the interface; this is essential as the mass spectrometer can detect and measure the ionized particles only.
As the analyte ions are created at atmospheric pressure in the interface, the process is called API (atmospheric pressure ionization), and the interface is known as the API source. ESI (Electrospray ionization) and APCI (atmospheric pressure chemical ionization) are the most widely used sources in Liquid Chromatography-Mass Spectrometry.
The analyte ions are drawn into the MS exposed to magnetic and electric fields. The flight paths of the ions are transformed by changing the applied fields, which confers their separation from each other based on their mass-to-charge values. The ions can be accumulated and detected by various mass detectors post-separation.The most commonly used mass detector is the electron multiplier. When the detached ions strike the surface of the electron-multiplier (a dynode), secondary electrons are released. These secondary electrons are multiplied by flowing them through a sequence of dynodes. The amplified current produced by the flow of the secondary electrons is measured and associated with the ion intensities in the mass spectrometer at any given prompt in time (Figure below). Rapid changes in the hardware are modulated and tracked by integrated software, with reported mass-to-charge ratios being based upon the electromagnetic fluctuation in each of the quadrupoles as ions reach the detector in order to differentiate mass-to-charge ratios.
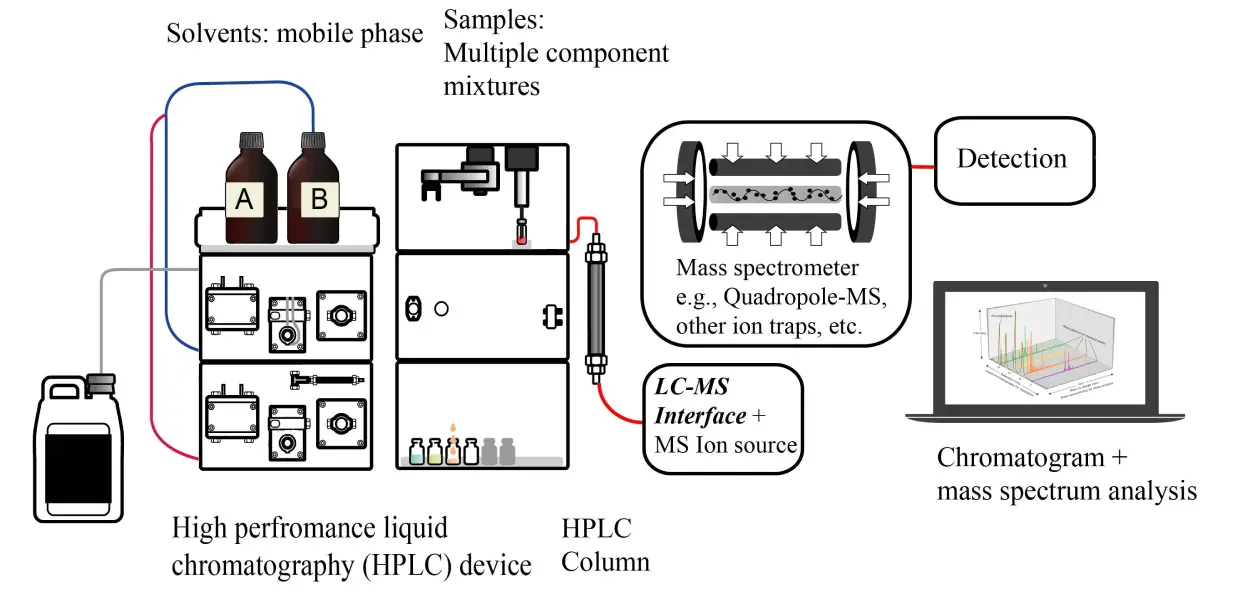
Figure : Example graphic diagram of an LC-MS setup. Source: Cwszot, Dagui 1929, Yassine Mrabet and Cas Ju reproduced under the Creative Commons CC0 1.0 Universal Public Domain Dedication license.
Plotting Mass Spectrometry for Liquid Chromatography data
The abundances of the ions quantified during the analysis of a sample by LC-MS are outlined as a total ion chromatogram (TIC). This plot shows the peak intensities of the analyte ions versus their retention time. Additionally, each point in the chromatogram is related with a mass spectrum. The mass spectrum displays the ion abundances versus the measured m/z values (Figure 2).
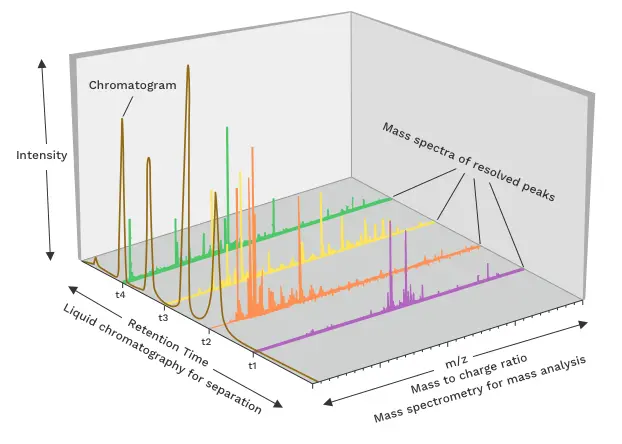
Figure 2: Example outcome plot from LC-MS analysis. Source: Daniel
Norena-Caro, reproduced under the Creative Commons CC0 1.0 Universal Public Domain Dedication license.
The mass spectrum of a compound provides information about the mass of the parent compound (from the m/z value of its ion). Additional structural information about test compounds may be inferred from the comparative abundances of spectral isotopic mass peak areas. The peak from the parent analyte (m/z or MRM) is used for its quantification.
The mass spectrometer (MS) can be operated in two modes, a) scan and b) selected ion monitoring (SIM). The scan mode is set to detect all the ions from low m/z to high m/z values within a specified period. This method is used when analyzing unknown samples or when there is no available information about the ions present in a sample, or to aid in the identification of impurities or metabolites in a sample mixture.. The mass spectrometer is set to measure specific m/z values when operating in SIM mode. SIM is the ideal mode of operation for accurately quantifying known compounds in a sample.
Combining LC with tandem MS
To improve sample identification and accurate quantification by coupling two mass analyzers operated in sequence. Dual quadrupole time-of-flight (TOF/TOF) and Triple quadrupole mass spectrometers (MS/MS) are the most frequently used tandem mass spectrometers. These configurations offer numerous options for sample analysis.
Precursor ion scan
The quadrupole one (Q1) is scanned over a mass range to select the precursor of a specific product ion m/z value is monitored in the last quadrupole (Q3).
Product ion scan
Precursor ion (Q1) transmits only the pre-defined precursor (m/z) to the collision cell. In contrast, Product ion (Q3) is scanned over a mass range to classify the fragments found under the experimental condition.
Neutral loss (NL)
Precursor ion (Q1) and Product ion (Q3) are scanned to identify all precursors that increase products by losing the similar uncharged (neutral) types from all the precursors. The NL value balances the scan range of Q3.
Selected reaction monitoring (SRM)
Q1 and Q3 are set to monitor actual m/z values for precursor and product ions. A Tandem Quadrupole Mass spectrometer (MS/MS) can monitor several precursor-to-product transitions of the identical and different analytes simultaneously. This mode is ideal for compound quantification due to its specificity and sensitivity.
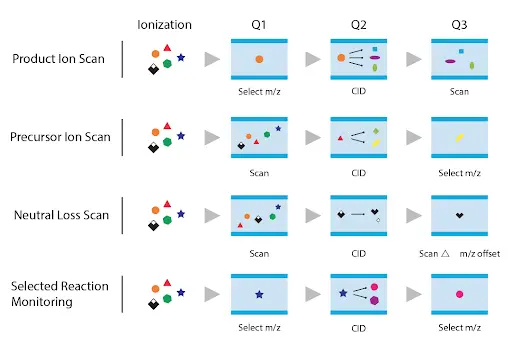
Figure 3: Modes of operation of MS/MS
LC-MS Analysis
LC-MS has been widely applied to analyze large proteins and small molecules in various matrices. A few instances of the real-world applications of this technology are: quantification of genotoxic impurities in active pharmaceutical ingredients, detection of twelve model compounds that represent specific classes of doping agents in athletes, such as anabolic agents and stimulants, in exhaled breath, quantification of drug metabolites in biological samples, detection of contaminants in food materials and dietary supplements, determination of alkylphenol ethoxylates (APEOs) in tannery sediments, quantification of toxins or environmental runoff in river water samples and personal care products in swimming pools, quantification of nucleotides and their derivatives in bacterial cells, or quantification of a proteome, for example as a rapid assay for the detection of SARS-CoV-2.
Strengths And Limitations of Liquid Chromatography Mass Spectrometry
Pros
LC-MS/MS has several analytical advantages, such as a more extensive linear dynamic range (typically), improved lower detection limits, higher accuracy, and precision because of the option to apply internal standards; the capability to quantify multiple analytes simultaneously; and the fact that it does not necessarily require immunological reagents. The last point can be especially critical because such reagents may be problematic to obtain or show large batch-to-batch variability, making evaluating results between laboratories over a more extended period difficult, if not impossible.
Cons
The disadvantages of LC-MS-MS include its high purchase, maintenance, and operational costs, limited sample throughput, lack of portability, and infrastructure needs for maintaining the power, gases, and exhaust for each system. Additionally, due to the need to maintain volatile mobile phase components going into the MS source, LC solvent system selections are limited and gases and plumbing used to operate the system must be of very high purity. Operating, troubleshooting, and maintaining instruments typically involves an expert team of trained analysts and engineers.
Common Issues with LC-MS
While LC-MS offers numerous advantages for trace analysis in complex matrices, several precautions have to be taken to overcome the following potential issues while using this technique.
Contamination
The selectivity, sensitivity, reproducibility, and resolution of analysis may be obstructed by contaminants, such as phthalates, metal ions, polyethylene glycol (PEG) or dosing vehicle components for in vivo PK assessments, water, slip agents, endogenous matrix components, and particles entering the system from various sources such as reagents and solvents, water used for preparing buffers, chemicals leaching from glassware, microcentrifuge tubes, inlet filters, solvent lines, instrument parts, such as pump seals, the sample itself, gases used for desolvation of the eluent in the source and the collision cell.
These contaminants can inhibit the analysis by directly overlapping, suppressing, or enhancing the ionization of analyte(s) in the source, forming adducts with the analytes or altering ionization potentials, masking the analyte peaks, changing signal intensities, or appearing as ghost peaks in the chromatograms, making the baseline noisy, fouling the system, causing failure to meet analytical specifications, and contaminating the LC column, requiring frequent maintenance and replacement of parts.
To minimize contamination:
High-purity solvents, water, and reagents should be used to prepare mobile phases; freshly prepared mobile phases essential be used to minimize the unintended microbial contamination of the aqueous mobile phase and polymerization of acetonitrile (ACN).
The usage of detergents/soaps should be avoided to clean glassware as they can be hard to remove and cause interference during analysis. High purity gases and plumbing are required (e.g., commonly used nitrogen gas of purity > 99%, cleaned burnished brass and swagelok fittings). Nitrogen generators must be well-maintained, and the gas cylinder must be replaced when the pressure is reduced below the acceptable level. Analytes must be extracted from the matrix and chromatographic parameters enhanced to improve the resolution of analyte peaks from interfering peaks. Considerations should also be made if particular contaminants interfere with the assay. For example, if an analyte of interest overlaps a known phthalate (plasticizer), plastics may be avoided, or if interference is seen from a drug’s dosing vehicle, dilution or other techniques may be necessary to ensure there is no signal interference from components present in selected samples.
Matrix effects
While evaluating biological samples, other sample constituents can suppress or enhance the ionization of the analyte in the source. Analytes of interest should be isolated to minimize the matrix effect. Therefore, sample preparation is an essential prerequisite of LC-MS analysis. While this decreases matrix effects, it cannot be easy to extract only the analyte(s) from the matrix. The chromatographic parameters can be optimized to prevent the co-elution of interfering compounds. Preparation of standard solutions in an analyte-free matrix (matrix matching) also helps normalize the analysis for matrix effects. Known concentrations of isotopically-labeled internal standards (IS), which experience similar ionization suppression or enhancement, additionally compensate for matrix effects.
Carryover
Analyte peaks may appear at the analyte retention time in blank injections run after high concentration samples due to sample analytical carryover. Carryover typically presents in the autosampler or LC column. Carryover may be addressed by using needle, solvent line, and column cleaning procedures, such as repetitive blank injections, column conditioning, and increased needle washes with multiple solvents to confirm the sensitivity and accuracy of the analysis is sustained.
Sample loss
Adsorption of analytes influences the precision and accuracy of the assay. Analytes, such as DNA and proteins, may be diminished due to non-specific binding to lab consumables, such as the inner surface of centrifuge tubes. Analyte loss can be minimized using containers with low surface bonding, for example, silanized glass. Usage of the blocking agents also minimizes the analyte’s interaction with the containers’ inner surfaces.
Mobile phase buffer selection
As the column eluent has to be detached before mass spectrometry analysis, only volatile buffers such as ammonium acetate or ammonium formate, which will not precipitate in the source, can be used to prepare mobile phases. Volatile acids include formic and acetic acid, and volatile bases include ammonium hydroxide.
Maintenance
Perform routine maintenance of the mass spectrometer as per a pre-determined schedule based upon vendor specifications and local standard operating procedures to ensure the instrument’s accuracy, reproducibility, and trouble-free operation and minimize unplanned downtime.