What is bioavailability?
Bioavailability is a key indicator of drug absorption. It represents the administered dose fraction which achieves success in reaching the systemic circulation when administered orally or through any other extravascular dosing route. Intravenous dosing is considered 100% bioavailable since the drug is administered directly to the bloodstream, also termed the central compartment or systemic circulation. However, if a drug has some different route of administration, oral being most commonly employed, its bioavailability may be limited. For oral doses, bioavailability limitations are typically due to the first-pass metabolism produced by the liver as well as incomplete absorption in the gut. Thus, it holds its importance as an essential pharmacokinetic tool and contributes widely towards the calculation of dosage for the different routes of administrations.
Bioavailability is determined by measuring pharmacokinetics in subjects following intravenous doses for reference and the intending dosing route (i.e., oral). Blood, plasma, or serum concentrations are determined over time and AUCs (area under the concentration vs. time curve) are calculated for each dosing route. Bioavailability is then calculated as the ratio of the dose normalized AUCs from the extravascular and intravenous routes. Bioavailability may differ between species, so these studies are critical in both the preclinical and clinical stages of drug research and development. Clinical bioavailability studies contribute to bioequivalence determinations since equivalent bioavailabilities of the generic and reference products constitute bioequivalence.
In the case of an orally administered drug, the pharmaceutical drug product has to surpass several obstacles to be considered suitable for oral delivery. In short, a pharmaceutical drug substance must –
- Undergo a complete liberation from its pharmaceutical product form.
- Undergo dissolution within the gastrointestinal fluid.
- Escape the metabolism undergone by the intestinal flora.
- Undergo absorption through the wall of the intestine either through active or passive permeation.
- Escape the gut wall metabolism.
- Escape the intestinal lumen excretion produced by efflux transporters.
- Escape first pass liver metabolism ahead of reaching the systemic circulation actively.
Considerations for bioavailability studies
The following types of considerations are paramount for all bioavailability studies –
- Selection of subjects
- Design of the study
- Washout period (for crossover designs)
- Study design – single-dose vs. multiple doses
- Conditions necessary to perform studies
- Pharmacological effects produced by the drug metabolites
- Assay Methods
Selection of study subjects:
To select the study subjects, age, health, enzyme status relative to hepatic and renal functions, weight, and the number of the subjects to achieve statistical significance are taken into consideration. The study subjects selected should possess similar kinetics for avoiding all significant forms of study variations and represent a healthy or diseased population as required by the intended therapeutic activity of the drug.
Study Design:
Usually, a bioavailability study is accomplished through a crossover study design. Under this study, all the study subjects involved receive the drug candidate with a certain washout period between two administered doses, one intravenous and one extravascular. While under the Latin Square study design, each of the subjects involved receives each route of administrations only once. Under the Latin Square crossover study, every subject act as self-control for the research. It helps in reducing the variations between two different subjects.
Washout period:
The time duration between two different treatments is defined as the washout period. The washout period is essential for the complete elimination of the administered dose of a particular pharmaceutical product under consideration. Thereby, for studying elimination of around 99.99 percent of the administered dose of the drug, a minimum count of 5 elimination half-lives are required in between two treatments. The washout period varies based on the drug’s metabolism and excretion and can be estimated following a pharmacokinetic determinations following a single intravenous dose.
Single-dose vs multiple-dose studies:
Single-dose studies are typically employed for determinations of bioavailability and bioequivalence. Multiple dosage studies are employed to determine time and number of doses to achieve steady-state exposure for chronic or continuous treatments, and to determine whether metabolic induction or saturation occurs with continued dosing. Multiple dose studies are required in preclinical drug development toxicology studies in order to establish appropriate safety margins for continuous administrations.
Study designs:
To accomplish a clinical bioavailability study diligently, several patient factors may be reviewed for inclusion or exclusion of subjects based on the drug’s therapeutic activity or anticipated toxicology. Factors to consider may include gender, age, weight, disease state, diet, other prescription or illicit drug use, child bearing potential, or other relevant factors. Before commencing your review, it is essential to pre-set study conditions such as drug pre-administration, fasting period, and post-drug administration period. Preliminary tests to determine enzyme function, pregnancy, or other relevant health factors are typically required prior to enrolment to ensure patients will provide the required study results in a safe and efficient manner.
Pharmacological effects produced by drug metabolites:
The measurement of bioavailability is dependent on the concentration of the unchanged drug. For drugs which possess highly active metabolites, their systemic circulation concentration can adversely affect a drug’s therapeutic efficacy. Metabolite concentration and safety evaluations are required for drug metabolites achieving significant systemic exposure or that have significant therapeutic activity. The evaluation is especially crucial for oral drugs undergoing high first-pass mechanism during their pre-absorptive stage.
Factors influencing the bioavailability studies:
The bioavailability of drugs is dependent upon several chemical parameters, including its pKa values, hydrophobicity, and solubility. Bioavailability of drugs with poor gut dissolution or absorption may be improved through enteric coating, extended release delivery mechanisms, or other dose administration routes.
Formulation of the pharmaceutical product:
For immediate release products, the excipients used in formulating the pharmaceutical product and the methods involved in manufacturing may alter bioavailability. All modified release drug products are studied for their delayed-release, extended-release, and sustained release to determine effects on the bioavailability of the drugs. Other factors influencing systemic absorption may include:
Whether the pharmaceutical product is administered during a fed or a fasting state
The rate of emptying the subject’s gastric system.
Whether the pharmaceutical candidate interacts with other drugs or food
This is studied either through understanding the mechanisms of enzyme inhibition or enzyme induction with in vitro ADME studies, or with PK studies comparing exposure in fed vs. fasted states.
It is essential to understand that each of the factors contributing to the bioavailability of drugs can vary in between patients. For clinical trials, the inter-subject variability is taken into account for assessing bioavailability differences between patients, thereby ensuring predictable dosing margins and risk factors for the general population.
How does the intake of food influences the rate of drug absorption?
The food you take in influences your drug absorption rate in two ways – physiological interactions and physicochemical interactions –
| Physiological Interactions |
| Physiological Functions |
Possible Effect on Drug Absorption |
| Stomach emptying rate |
The overall absorption rate of a drug substance is influenced by gastric emptying. Absorption may be reduced due to instability at low pH or increased due to drug dissolution. |
| Intestinal motility |
The faster rate of drug dissolution and decrease in its overall diffusion contributes towards more and more drug absorption. Therefore, shorter transit time may cause inhibitory effects on the rate of drug absorption. |
| Splanchnic blood flow |
Splanchnic circulation describes the blood flow to abdominal gastrointestinal organs. The rate of drug absorption increases with a faster flow of blood. It produces variable effects on the first pass mechanism exhibited by the liver since the portal vein delivers orally absorbed compounds to the liver prior to entry to systemic circulation. |
| Bile secretion |
Bile secretion may either increase drug absorption due to a faster dissolution rate or decrease it due to complexation or biliary excretion. |
| Acid Secretion |
Increase in overall basic drug absorption for compounds that are acid stable. The presence of acid-secretion may lower the absorption rate of all those compounds that are acid-labile. |
| Enzyme secretion |
Increase or decrease in the rate of drug absorption, which is highly influenced by the metabolic characteristics of a drug compound. |
| Active process of drug absorption |
The active process of drug absorption may be lowered due to competitive inhibition from other drugs or food products. |
| Physicochemical Interactions |
| Interactions |
Possible effects on drug absorption |
| Volume of fluid |
A decrease in the absorption rate of large fluid volumes reduces the drug concentration gradient. Features such as faster dissolution, exposure of drug molecules to greater GI surface area, and osmotic effects may cause an increases in absorption efficiency. |
| Food and its components |
Features such as adsorption, chelation, absorption, and physical blockades may introduce a decrease in the overall rate of drug absorption. However, an increase in overall diet-induced drug solubility or lipophilicity may increase the overall rate of drug absorption. Absorption variability may be influenced by changes in pH values. |
What is a bioequivalence study?
A pharmacokinetic term, bioequivalence assesses the in vivo biological exposure equivalence of two different proprietary drug preparations. Two drug products are considered to be bioequivalent when they produce similar systemic concentrations and pharmacologic effects. In fact, both bioavailability and bioequivalence studies focus on the release of the drug substance from a finished pharmaceutical drug product and its subsequent absorption into systemic circulation.
Considerations for performing a bioequivalence study
Usually, a crossover design is generally employed in conducting BE studies. To perform these studies with higher relevance, intra-subject variability is taken into consideration. In the case of a parallel crossover study design being employed in BE evaluation, emphasis should rely more on total subject comparability and variability. The total subject variability should be include greater inter-subject variability than intra-subject variability.
General considerations
The FDA and other global regulatory agencies usually focus more on the rate and extent of absorption of an active drug moiety from its pharmaceutical end product to reaching its targeted site of action. To accomplish in vivo studies more efficiently, it is essential to determine PK bioanalysis with calibrators and controls using matching biological matrix (plasma, blood, or serum) for indicating the release of the active drug substance from its final product into systemic circulation.
Pilot Study
Pilot studies are often employed to initially validate the analytical methodology, determine the size of the sample required for accurate and precise analyses, assess PK variability, optimize the time course and procedure for collection of the samples, and determine the appropriate time length for washout periods.
Full-scale study
Modified release and immediate release dosage forms are usually tested through non-replicate crossover study designs. However, presently, most of the clinical research institutes employ replicate study designs for performing their bioequivalence studies. Replicated crossover study designs provide an estimation of inter-subject variance both for reference and test products.
Study population
Subjects included under the BA and BE studies should be more than 18 years of age and should be able to give informed consent. It is a regulatory requirement to accomplish both the BA and BE studies in healthy volunteers if it is possible to administer the drug candidate in a healthy population.
Single and Multiple-dose testing
21 CFR 320.27 provides regulations on designing in vivo multiple-dose BA studies. The regulatory requirements for undertaking such studies are described as follows:
- There can be a difference in the absorption rate, but there should not be any difference in the extent of drug absorption.
- The bioavailability varies significantly between two different subjects involved in the pharmacokinetic study.
- The concentration of the active pharmaceutical ingredient or any of its active metabolites contained in the blood as determined by the single-dose study is too low to be accurately determined by analytical methods.
- The drug product employed in the study has to be present in an extended dosage form.
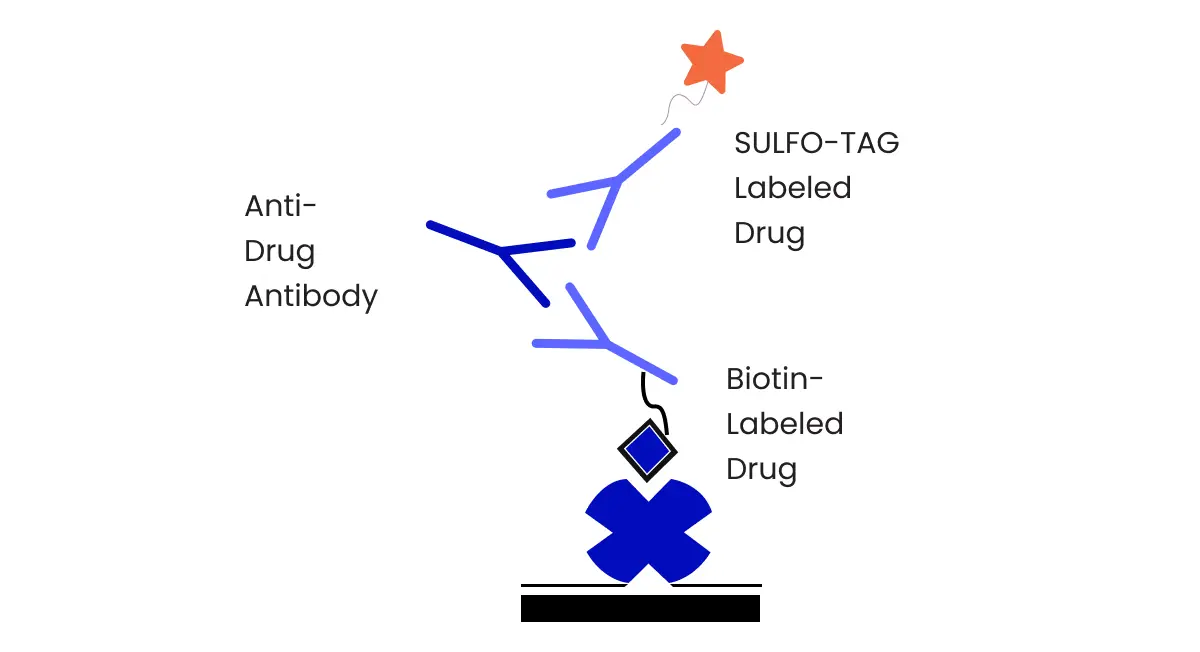
Bioanalytical Methodology
All the bioanalytical methods employed in BA and BE studies should be precise, accurate, sensitive, specific, and produce reproducible results. Preclinical toxicology and clinical studies require validated analytical methods to insure these factors along with compound stability.
Fasting or Fed administrations
The BA or BE studies should be accomplished under fasting conditions, i.e., after overnight fasting of at least 10 hours. The exception to be observed for fasting administration a lack of tolerability or any adverse reactions. If such cases are found, it may become essential for clinical researchers to conduct fed studies for comparison. Several marketed drugs are have labels indicating the requirement to take with food based on these observations.
Measurable moieties
The biological fluids that are collected throughout BA and BE studies should be evaluated to measure the concentrations of active pharmaceutical moieties and not their metabolites. The only exception of allowing the measurement of the metabolic intermediates is that the active pharmaceutical substance is present in quite low amounts that can be determined in the plasma, blood, or serum. In the case of prodrugs, both the prodrug and the active moieties should be measured.
Pharmacokinetic measurement for systemic exposure
To determine the complete pharmacokinetic analysis of systemic exposure, the clinical researchers must focus on designing studies with appropriate dosing and sample collection time courses to determine systemic absorption, distribution, and clearance through metabolism and excretion.
Parent drug versus metabolites
The basis of the bioequivalence studies relies on adequate sampling of biological fluids for determining drug presence. The concentration of the parent drug should always be measured in these biological fluids until the compound becomes undetectable in systemic circulation. In general, it is only the parent drug concentration that is determined than rather than any intermediate metabolites. This is so because the parent drug concentration profile more directly reflects changes in formulation performance than what is seen in the case of intermediate metabolites.
First Point Cmax
The concentration profile for the drug candidates assesses both the absorption and elimination of the drug compound present in the biological fluid. When the first measured time point following dosing for an extravascular dose represents the highest point in a concentration-time curve, it may indicate the need for earlier and more frequent collections in order to appropriately estimate the compound’s true Cmax. Proper time courses can be determined from pilot studies to provide the most reliable estimations in pivotal bioavailability and bioequivalence studies.
Effects produced by alcohol on drug products belonging to the modified release drug categories –
During protocol and study design generation, appropriate sampling schedules need to encompass the desired release characteristics of the drug candidate. If alterations in drug absorption, exposure, or efficacy are expected in the presence of alcohol or other drugs, the product label must reflect appropriate warnings regarding restrictions and potentials for drug-drug interactions. These interactions are typically predicted via in vitro metabolic inhibition, substrate, genetic polymorphism, and induction studies.
Endogenous compounds –
Endogenous compounds are present within our body, either due to being processed by the human body or being a part of the regular human diet. They include proteins, peptides, amino acids, nucleic acids, antibodies, and many other moieties. In cases where endogenous compounds are used as active drugs, endogenous levels may interfere with absorption of the drug and will require additional efforts to clear endogenous materials from calibrators and controls during sample bioanalysis. Additionally, diurnal variations in endogenous exposure may complicate PK determinations.
FDA regulations on the site requirements to perform bioavailability and bioequivalence studies
The following criteria are required at the clinical study sites –
- Designated rooms to volunteer subject registration and cleaning
- Designated places for volunteer recreational areas
- Designated places for informed volunteers for obtaining informed consent
- Designated ancillary areas for the study volunteers
- An area designated for pharmacy requirements
- Areas designated for restricted pharmaceutical operations
- Rooms designated for dosing and sample collection of the drugs under investigation
- Designated area for processing and storing samples
Types of equivalents available in the market
Pharmaceutical equivalents –
Two drug products are said to be pharmaceutically equivalent when both the drugs contain equal amounts of the active pharmaceutical ingredient. Pharmaceutically equivalent products are established to be identical dosage forms. While it is not necessary that they have to include the same inactive ingredients, bioequivalence of the active pharmaceutical ingredient must be demonstrated.
Pharmaceutical alternatives –
Pharmaceutical alternatives are defined as identical therapeutic moieties or their precursors. However, they may include structural alterations including prodrug moieties such as phosphates, esters, or other biologically labile functional groups, or different salt forms. The dosage form and concentration of the final pharmaceutical product may differ.
Bioequivalence –Bioequivalence is defined as pharmaceutical equivalents or alternatives of a therapeutic drug product only when –
- Both the test and reference drug products are administered in the same amounts at the same defined molar doses.
- Produces equal rate and extent of drug absorption.
- They are not statistically or significantly different from all their reference products available on the market.
Therapeutic Equivalence –
- Two drugs are said to be therapeutically equivalent only when they contain the same active substance or therapeutic moiety; and
- Are clinically potent enough to exhibit the same efficacy level and drug safety profiling
Exclusion criteria for bioequivalence studies –
While they may vary based upon therapeutic area and preliminary toxicological assessments, the following rules are typically considered as exclusions to bioequivalence studies:
- Exhibit potent allergy to test drugs
- Exhibited liver or kidney dysfunction.
- Suffered from jaundice in the past 6 months.
- Suffered from chronic disorders such as arthritis or asthma.
- Suffered from psychiatric illness.
Types of products requiring bioequivalence studies –
Unless justified, the bioequivalence studies are quite crucial for the following NDA’s –
- Immediate-release capsules or tablets categorized under the orally administered category.
- Modified release capsules or tablets categorized under orally administered drugs.
- Systemic action dependent transdermal patches.
- Suspensions and solutions categorized under orally administered drugs.
- Complex parenteral solutions such as micelle forming formulations, liposomal forming formulations, and emulsions.
- Systemic action dependent non-oral immediate release dosage forms.
When are bioequivalence studies not required?
The following new pharmaceutical drug products do not require any need to undergo bioequivalence studies –
- Therapeutic substances based products which are not absorbed either through local absorption or systemic absorption.
- Medicinal gases
- Nebuliser solutions
- Simple aqueous powders or intravenous solutions explicitly designed for reconstitution.
- Biosimilars
- Local action based nasal sprays
- Vaccines
Challenges before Clinical Endpoint Studies –
- Clinical endpoints are more variable concerning PK studies; however, they must justify the established BE limits.
- Clinical endpoint studies require a reasonable number of patients to establish statistical significance.
- The duration of the study may range up to several weeks, depending on the approval received for labelling.
- Clinical studies are costly to be conducted.
- Clinical studies may exhibit more concerns related to safety than PK studies.
Why are Bioequivalence studies importance in drug development and research
Bioequivalence studies are crucial for the reasons mentioned below –
- Evaluating relative or absolute bioavailability of dosage forms upon being compared with reference dosage forms.
- Performing dose proportionality studies for determining the linearity of the bioavailability parameters over the proposed dosage range.
- Variations within or between subjects.
- Performing intervention studies for examining effects both of food and concomitant medications.
- Performing dosage form proportionality studies for determining an equipotent drug treatment being administered at different dose strengths producing equivalent bioavailability of drugs.
- The requirement of bioequivalence studies for ascertaining changes during formulation or manufacturing processes.
Conclusion
Adherence to the FDA protocols and guidelines will ensure that the bioavailability and bioequivalence tests conducted are accurate and productive. The two studies are integral to drug development, testing and research as they expedite the process and reduce the overall costs of a generic drug reaching the market.